Chinese man loses two front teeth trying to eat raw corn on the cob off an
Figure 3. This view of the scandium carbide, Sc3C4, structure emphasizes the bonding in the carbon subunits. Note the presence of C3, C2 and isolated C fragments.
Ask an American chemistry graduate student, "Tell me what you know about carbides," and here's what I guess you'd get as an answer: "There's calcium carbide, CaC2, and I've heard of those long chains of carbon in John Gladysz's organometallic molecules, and, yes, some transition metal carbide clusters, for instance the iron carbide carbonyl Fe5C(CO)15 [Figure 1]."
Finally, a personal view of the importance of the carbides. It's not in their economic value. Nor even in the simple to strange beauty of their structures, the paean to complexity they silently sing. No, their message is a spiritual one. The carbides are inorganic. Yet, ever so clearly, they are also organic—how else shall we think of those C-C bonds in many of them? Metal carbides are a bridge, the inherent link nature itself shows us, as we, in the simplicity of our minds, pigeonhole into one or another human-made category—organic, inorganic—the multifarious manifestations of one complex world.
In fact, the few hundred carbides known are a most remarkable and, as I will argue, inspiring group of compounds. First a brief on their macroscopic properties: These are all solid compounds, ceramic, sometimes metallic, often hard, often with high melting temperatures. They may be sensitive to moisture (Can anyone who has experienced it forget the smell of wet technical grade CaC2?), but often are very resistant to water and air (those WC drilling bits). Their compositions may be simple—say, WC or NbC (niobium carbide)—or complex—Sc3C4 (scandium carbide) or Er10Ru10C19 (an erbium ruthenium carbide).
As more carbon-rich carbides are synthesized, I think we will see other forms of C in them, and not only C, C2 and C3. In fact, I'm sure that little chunks of carbon will be made in solids in shapes that are so far unknown for carbon. Why? Well, every environment for the growth of an element (here carbon) creates its own "conditions of stability"—free in the gas phase or solution, on the surface of a metal, in the metal's interior, at an interface. This is how I think about the fact that buckminsterfullerene, C60, reached macroscopic stability—in the special reaction conditions of a carbon arc in a helium atmosphere. For most forms of carbon, survival after formation is guaranteed by the high barriers for breaking strong C-C bonds, even in strange, highly strained geometries. Based on what we know about the unusual geometrics of solid state borides (boron is next to carbon in the Periodic Table), it's just a matter of time until we find weird C-C bonding in carbides.
With a long-standing reputation for quality and durability, Ruwag offers the best quality products for every type of project. We have all the bits you need under one roof. Contact us today for more information on our world-class drill bits for concrete.
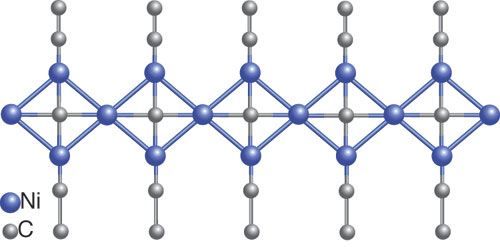
Retreating just a bit from this vertiginous edge of complexity, consider Ca4Ni3C5, also made by the Jeitschko group at Münster in Germany. (Wolfgang Jeitschko is responsible for a great fraction of the carbides we have.) Figure 4 shows the one-dimensional Ni3C58– ribbon cut out of the structure. It has vertex-sharing nickel squares, with C2 wingtips. And smack in the middle of each square in this piecewise organic molecule—a square-planar (not tetrahedral) carbon!
Drill bits for concrete have been specially designed to drill into extremely hard masonry. It is essential to use the correct bits for projects involving concrete. Using general-purpose bits or other types of bits made for softer masonry can damage your bits. You also run the risk of damaging your power tools or even causing injury, as it is very difficult to control the drill when the wrong bits are used to try and drill into hardened materials.

In YCoC there is a CoC3– organometallic polymer, multiply bonded between Co and C, judging from that short distance. The polymer is trapped in the solid state. A theoretical chemist wishing to provoke his uppity experimental colleagues (that's me) says, "Get it out of there, solubilize that polymer, give it—in a solvent—a better Lewis acid partner than Y3+." Of course, they won't get it out. But this structure should be an inspiration to molecular organometallic chemists making rodlike polymers; they're possible, waiting to be made outside the solid.
Whether you're starting a home renovation project or an industrial build, there are a few different types of drill bits for concrete. In this guide, we share some of the best bits you will need for drilling into concrete. Keep reading to find out more.
The final and rare mode of C-C bonding in carbides is a C3 unit. It occurs in Ca3Cl2C3, but I'd rather show you the phantasmagoric Sc3C4 structure of Pöttgen and Jeitschko (Figure 3). Like a smorgasbord of carbon forms in carbides, it has everything—C and C2 and C3 units in a complex arrangement whose repeat unit is Sc30C40, containing 12 C, 2 C2 and 8 C3. It is as if the metal atoms had torn apart a graphite lattice, forming little islands of organic matter in the metal.

The tips of masonry bits are very similar to twist bits. They have a similar type of shaft and shank. They have a profiled tip with flutes that help to remove dust as the drill hammers into the concrete. The unique shape of this tip helps the drilling process. It makes it easier for the bit to break into hardened masonry such as concrete using a hammer drill. Turbo concrete bits have rotary hammer grade centering tips that make them ideal for fast drilling. Industrial concrete bits have profiled tips that allow for heavy drilling.
The realities: Calcium carbide, CaC2, remains a major commercial chemical, though I suspect that few connect that molecule to the name of the one-time chemical giant, Union Carbide Corporation, now a subsidiary of Dow; WC—no, not the loo but tungsten carbide—in drilling bits and in "cemented" carbides is a major industry. Looming above these is steel, of weapons and at the core of large constructions. Steel is hardly uncommon, but I'd bet there is nary a word about this alloy of iron and carbon in the education of our students. Steel is a solid solution of carbon in iron mixed with compounds such as iron carbide, Fe3C, and Hägg's carbide, Fe5C2. Besides these three major players—CaC2, WC and the carbides in steel—many other metal carbides have been made, more for curiosity than for profit.
To discuss our articles or comment on them, please share them and tag American Scientist on social media platforms. Here are links to our profiles on Twitter, Facebook, and LinkedIn.
Figure 1. This carbide made of iron, carbon and oxygen, Fe5C(CO)15, is a molecular carbide cluster. In contrast, most carbides have extended structures that cannot be divided into discrete molecules.
The lacunae in the student's answer—and professors will do no better—tell us much about education and fashions in science. I see in the answer (a) the triumph of molecular chemistry in the past century, so, for example, more people know the later and rarer discrete Fe5C(CO)15 cluster than the extended Fe3C structure, and (b) the love affair people—subspecies chemists—have with simplicity, so both teachers and students would rather deal with a simple discrete molecule rather than the difficult to explain lanthanum nickel carbide, La2Ni5C3, or, God forbid, messy steel.
Choosing the right size bit is always important, whatever material you are drilling. The size will determine the depth you can drill. Ruwag offers turbo and industrial concrete bits in a selection of sizes. Turbo concrete bits range in size from 5x85mm to 14x150mm. Industrial concrete bits range from 3x60mm to 16x150mm. We offer sets for both types of bits, with a selection of sizes. This is the best way to get the right size for the job at hand.
If you’re looking for extremely powerful bits that are ideal for concrete and other hardened materials, SDS bits are an excellent choice. These bits are tough enough to break through reinforced concrete. Our selection includes SDS-Max and SDS-Plus bits. SDS-Max bits are larger and thicker, while SDS-Plus bits have a slimmer width. These bits can only be used in SDS drills unless you use an SDS chuck made for hammer drills.
We would not know many of these carbides if it were not for the fruitful synthetic and structural studies of Wolfgang Jeitschko. I am grateful to him for inspiration, to Hans-Jürgen Meyer for his comments and to M. M. Balakrishnarajan for his assistance with producing the drawings.
Drilling into concrete is challenging and tools often overheat. Concrete drill bits have carbide tips that allow them to drill into concrete and stone. This material is made by combining carbon and tungsten, resulting in a dense, hard metal. It has a high melting point, which means that bits featuring carbide tips can withstand a very high level of heat. This, in turn, helps to prevent over-heating and the damage it can causing during drilling. Always choose bits with carbide tips so that you are able to get the best results without wear and tear damaging your bits.
There are many carbides that contain two linked carbon units. CaC2 is one, a simple structure in which the C2 units are close to C22–, like an acetylene (HCCH) with the two hydrogens ripped off as protons. Consistently, the C-C bond length in the C2 units of CaC2 is acetylenic, 1.19 angstroms. There's a drawing of the atomic positions in calcium carbide in another article I've written (Marginalia, July–August 1995). C-C bond lengths in the C2 units of other carbides range remarkably, from 1.19 to 1.48 angstroms, nearly matching the range of C-C bond lengths in organic molecules, from a triple bond to a single bond. This is not an accident.
Incidentally, if you ask an inorganic chemist about the date of the first synthesis of molecules with a metal atom in a trigonal prismatic environment, the likely answer will be approximately 1965. But the WC structure has been known since 1928; it clearly has a trigonal prismatic coordinated metal atom, with W-C distances typical of those in organometallic molecules. That the molecular inorganic chemist doesn't point to WC (or to MoS2—molybdenum sulfide—another structure with trigonal prismatic coordination of a metal, given to us by Linus Pauling in one of his early papers in 1923) illustrates the separation, persistent even as it is illogical, of molecular and extended inorganic chemistry over most of the 20th century. Talk of two cultures!
Figure 2. These structures of niobium carbide (left) and tungsten carbide (right) do not emphasize the Nb-Nb and W-W bond distances. Note the octahedra of Nb around C and octahedra of C around Nb at left. Similarly, one sees at right trigonal prisms of W around C and trigonal prisms of C around W.
Consider still another Jeitschko structure, made of yttrium, cobalt and carbon—YCoC (Figure 5). Through it run infinite [CoC]3– needles, with a cobalt-carbon separation that is tied (with a discrete molecule) for the world's record for shortest Co-C distance (1.85 angstroms). The Co-C separation in the naturally occurring molecule vitamin B12 is substantially longer.
Now to their microscopic structure. In the carbides known, carbon appears so far in only three forms. Most common are isolated carbon atoms, probably closer to being negatively charged ions. Figure 2, for instance, shows the structure of NbC—a layering of niobium and carbon, with an octahedron of niobiums around each carbon and an octahedron of carbons around each niobium. Does that sound familiar? These words describe the geometrical structure of rock salt or NaCl (sodium chloride), seen in our view of NbC from an atypical angle. The distances between niobium atoms are a little longer than in niobium metal; the Nb-C distances are similar to those in discrete molecular NbC organometallic compounds. And there are no C-C bonds. Figure 2 also shows the WC structure, in which each carbon is surrounded by a trigonal prism of tungsten atoms and each tungsten by a trigonal prism of carbons. Interesting that the two structures should differ so much; I've written a paper with Sunil Wijeyesekera teasing out an explanation for the different coordination preferences of NbC and WC. Cementite, Fe3C (an important form of carbon in steel), is a variant of the WC structure. To get to it, imagine removing two-thirds of the carbon atoms and then collapsing the metal lattice, albeit in a complex way.
Figure 4. Infinite nickel carbide (Ni3C5) chain is cut out (and idealized) from the calcium nickel carbide, Ca3Ni3C5, structure. Note the carbon atoms in the middle of vertex-sharing Ni squares.
There are a few things that you will need to consider when choosing drill bits for concrete. Here are the most important factors to keep in mind.
Figure 5. A piece of the yttrium cobalt carbide, YCoC, structure appears with one CoC chain elongated to show more clearly its polymeric rodlike shape; the other CoC chains are no different from this one.




 18581906093
18581906093